2.1 THE NATURE OF MATTER
Every thing is made up of atoms. Atom is the basic unit matter. Scientists calculated that if 100 million atoms line up together, it would only make 1 centimeter. Even though atoms are extremely small, they are still make up of 3 subatomic particles.
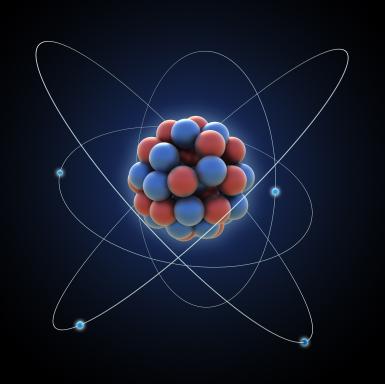
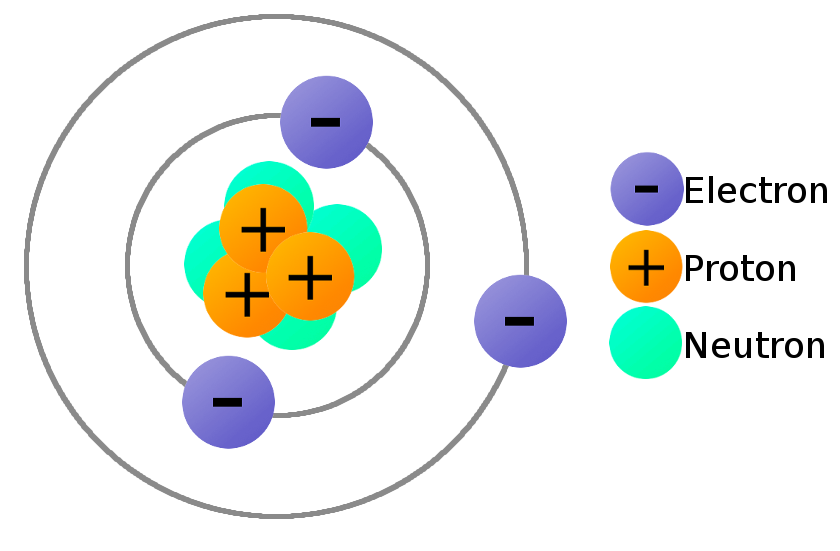
The three subatomic are the neutrons, protons, and electrons. The center of the atom (look at the image above and below) is called the nucleus. The nucleus has a strong force that binds the protons and neutrons together. The proton is the positively charged particle (+). The neutron is the neutral particle. The electron revolves around the atom and is a negatively charged particle (-). The electron are attracted to positively charge nucleus but still remain outside the nucleus because of the energy of motion. Atoms are neutral because they have equal electrons and protons.
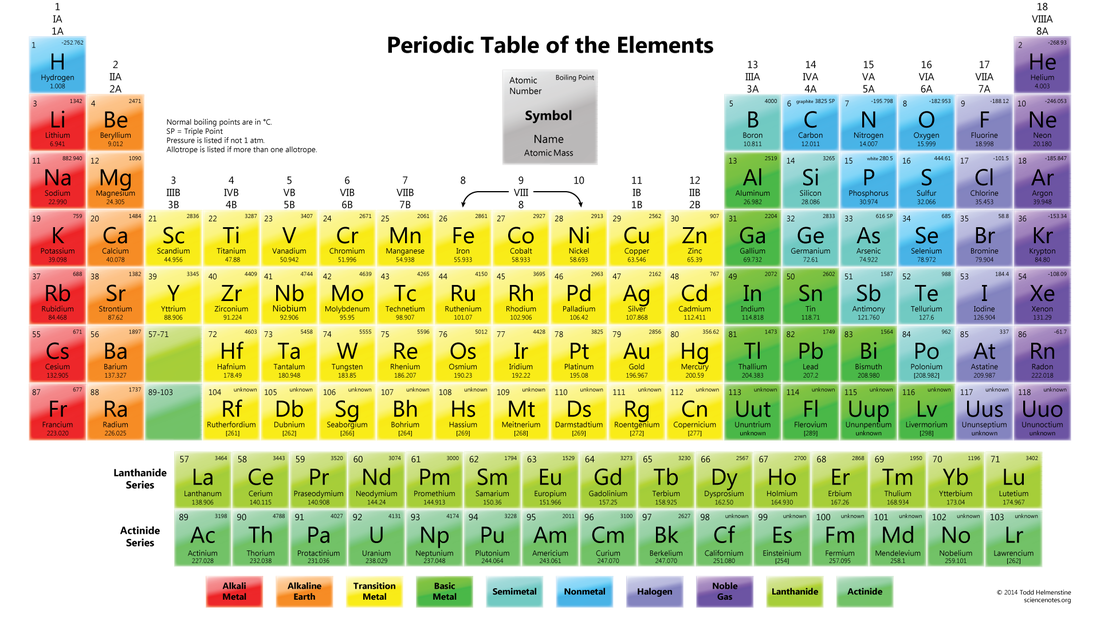
There are 100 elements are know but only about two dozen are commonly found in living organisms. A chemical element is a pure substance that consists of one type of atom. such as carbon (C) or hydrogen (H). Elements will have an atomic number. Example: Carbon's atomic number is 6 which means it have six protons and six electrons. All of these elements can be found in the Periodic Table.
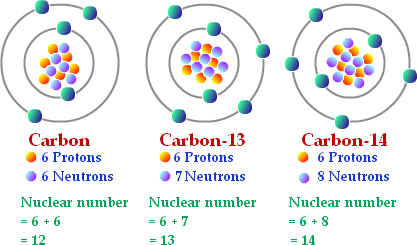
Atoms of an element can have different numbers of neutrons. Atoms of the same element that differ in the number of neutrons they contain are known as isotopes. The sum of protons and neutrons in the nucleus of an atom is called its mass number.The weighted average of the masses of an element's isotopes is called its atomic mass. But because they have the same number of electrons, all isotopes of an element have the same chemical properties. Some isotopes are radioactive which means that their nuclei are unstable and break down at a constant rate over time. These isotopes can be dangerous however, but it can also be helpful due to it helping geologists determining the ages of rock and fossils by analyzing the isotopes found in them.
Most elements can be found combined with other compounds. This is called compound. A chemical compound is a substance formed by the chemical combination of two or more elements in definite proportions. Some of these compounds are H2O (water) or NaCl (table salt).
The main types of chemical bonds are ionic bonds and covalent bonds. An ionic bonds is formed when one or more electron are transferred from one atom to another. If the first atom transferred an electron to the second atom, it then becomes positive and the other becomes negative. These positively and negatively charged atoms are known as ions. A covalent bond forms when electrons are shared between atoms. When atoms share two electrons, the bond is called a single covalent bond. The most bond two atom can have is a triple covalent bond. The structure that results when atoms are joined together are called a molecule. A molecule is the smallest unit of most compounds.
Atoms if different element do not have the same ability to attract electrons. Van der Waals forces is how chemists call intermolecular forces of attraction (Example: A gecko's feet). Atoms made everything in the universe. The subatomic particles that make up atoms are protons, neutrons, and electrons. Even though Van der Waals forces is not as strong as the other two bonds, but it can hold molecules together, especially the molecules that are large.
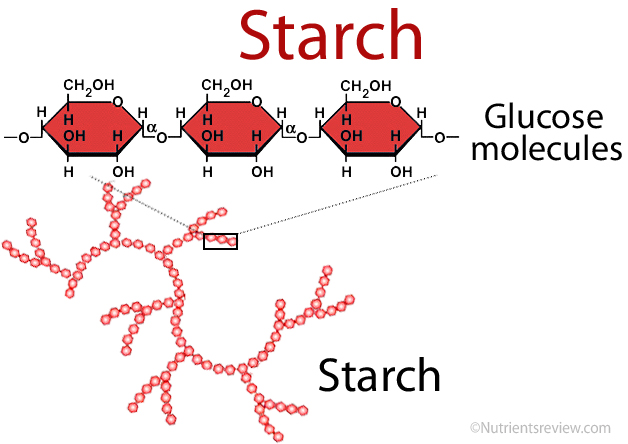
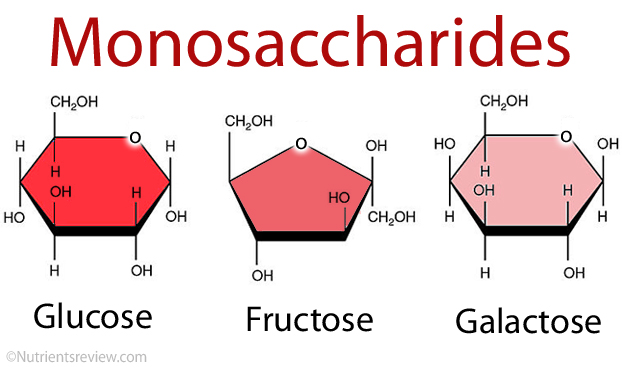
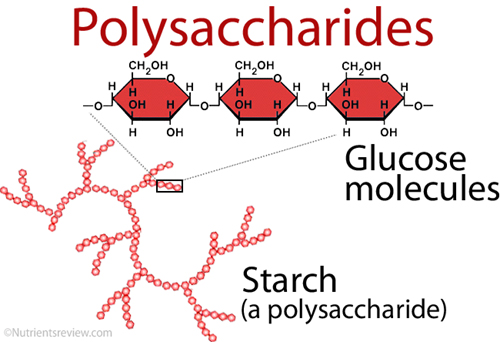
Monosaccharides are single sugar molecules. Besides sugar monosaccharides include latacose which is milk and glucose which can be found in fruits. The large macromolecules formed from monosaccharides are known as polysaccharides. Animals store excess sugar in a polysaccharide called glycogen, or animal starch. When the blood runs low, glycogen is released from your liver and be an energy for muscle contraction (movement). Plants use polysaccharide a bit different. They store excess sugar called plant starch to help them be tough and flexible.
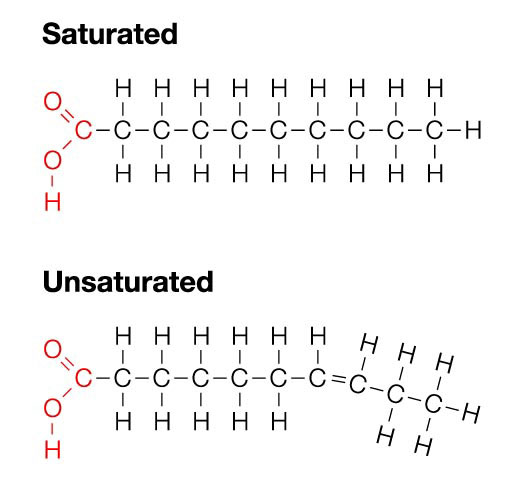
Lipids are made mostly from carbon and hydrogen atoms (fats, oils, and waxes). Lipids can be used to store energy. Some lipids are important parts of biological membranes and waterproof covering. Steriods are also limpids and can be use as chemical messengers. The term saturated is used because the fatty acids contain the maximum possible of hydrogen atoms. If there is at least one carbon-carbon double bond in a fatty acid, the fatty acid is said to be unsaturated.
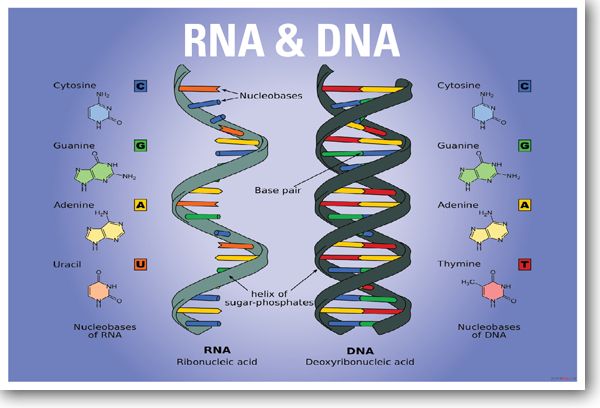
Nucliec acids are macromolecules, containining hydrogen, oxygen, nitrogen, carbon, and phosphorus. Nucleotides consist of three parts: a 5-carbon sugar, a phosphate, and a nitrogenous base. Nucleic acids store and transmit hereditary, or genetic information. There are two kinds of nucleic acids: Ribonucleic acid (RNA) and Dexoyribonucleic acid (DNA).
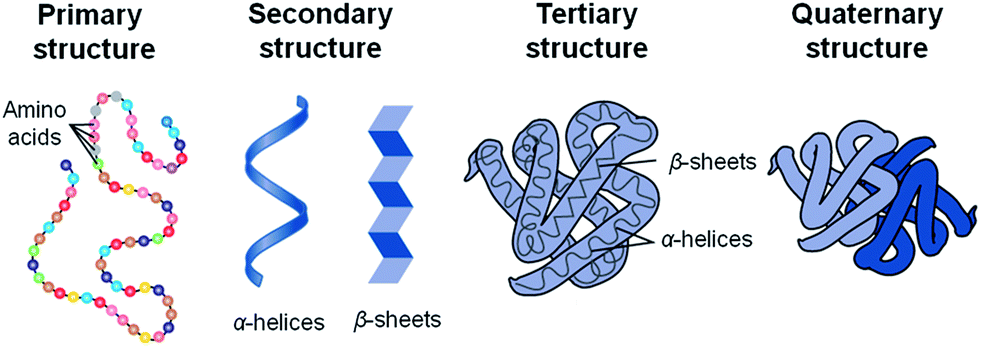
Proteins are macromolecules that contain nitrogen as wall as carbon hydrogen and oxygen. Proteins are polymers of molecules called amino acids. Some proteins control the rate of reactions and regulate cell processes. Some are used to from bones and muscles. Other transport substances into or out of cells or help to fight disease.
ALL PICTURES ARE FROM GOOGLE IMAGES.